Stoichiometry and Limiting Reactants Activity
Abstract
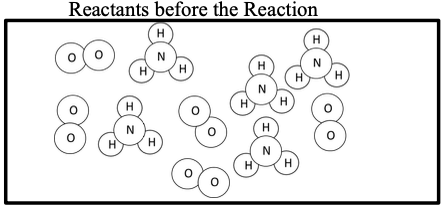
This activity aims to develop students understanding of limiting reactant stoichiometry at the particulate level in addition to manipulating reaction stoichiometric amounts mathematically. The activity starts with a sticky note activity building and reacting molecules until no further products can be formed. Students discover that although they started with a 1:1 reactant system, not all of the reactants could be used to form products. Students are asked to apply and define the following terms: make/produce/yield, use, excess, and limit. Students then relate the balanced chemical equation to the amounts that reacted in the sticky note exercise. The intent is to get students to define a balanced equation in terms of ideal, lowest whole number ratio of reactant to products instead of trying to rewrite a balanced equation based on actual amounts used in a reaction. Students work through molecule to molecule and mole to mole relationships in a reaction with excess reactants, once again requiring them to apply the earlier defined terms. Students then are guided to calculate amounts in a reaction with excess reactant to discover that conservation of mass is still followed although some of the mass is still as unreacted reactant. The final part of this activity applies these concepts by starting with gram amounts of reactants but once again asks students to apply the earlier defined terms.
Level: Undergraduate or Advanced High School
Setting: Classroom
Activity Type: Learning Cycle
Discipline: Chemistry
Course: General Chemistry
Keywords: balanced chemical equations, stoichiometry, particulate nature of matter
Downloads
Published
How to Cite
Issue
Section
License
Copyright of this work and the permissions granted to users of the PAC are defined in the PAC Activity User License.

