Stoichiometry
Abstract
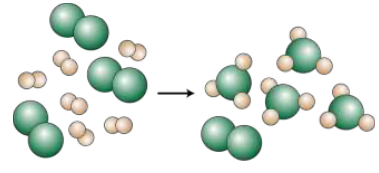
Balanced chemical equations are symbolic descriptions of a chemical reaction and the relative proportions of each reactant and product involved are indicated by the coefficients. The mole ratios from the balanced chemical equation can be used in stoichiometry calculations to determine the limiting reagent and theoretical yield.
After completing this activity, students will be able to determine the stoichiometric relations, or mole ratio, or reactants and products from a balanced chemical equation; determine the limiting reagent; and, use mole ratio and limiting reagent to calculate the theoretical yield.
Level: Undergraduate
Environment: Classroom
Activity Type: Learning Cycle
Discipline: Chemistry
Course: General Chemistry / Introductory Chemistry
Keywords: Stoichiometry, mole ratio, limiting reactant, theoretical yield
Downloads
Published
How to Cite
Issue
Section
License
Copyright of this work and the permissions granted to users of the PAC are defined in the PAC Activity User License.

